Amblyomma spp.
Amblyomma hebraeum | Amblyomma variegatum | Vector-based epidemiology of heartwater
Amblyomma hebraeum – the bont tick
It is known as a bont tick because of its bright contrasting colours. It transmits Ehrlichia ruminantium and Theileria mutans.
The distribution of the tick is confined to South Africa, Swaziland, Botswana, Zimbabwe and southern Mozambique, and does not overlap with that of A. gemma.

Amblyomma hebraeum |
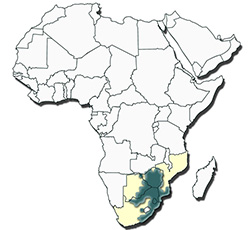
Distribution of Amblyomma hebraeum |
Adults feed on cattle, sheep, goats and large wild ruminants, particularly giraffes (Giraffa camelopardalis), African buffalo (Syncerus caffer) and eland (Taurotragus oryx), and also on warthogs (Phacochoerus africanus) and black (Diceros bicornis) and white rhinoceroses (Ceratotherium simum). Immatures infest the same hosts as the adults but also small antelopes, helmeted guineafowls (Numida meleagris), scrub hares (Lepus saxatilis) and leopard tortoises (Geochelone pardalis). The immature stages of this tick do not infest rodents (rat, mice, squirrels, gerbils), if they do they seem unable to engorge and usually die. The adults prefer the hairless areas under the tail, in the lower perineal region, on the udder and testes, around the prepuce and in the axilla of cattle, as well as around the feet of sheep and goats. The larvae are found on the feet, legs and on the muzzle, the nymphs attach on the feet, legs, groin, sternum and neck.
Amblyomma hebraeum is a three-host tick. The adults and nymphs are “hunters”, scuttling along the ground when a suitable host is in the vicinity, particularly so if the host is already infested with adult male ticks excreting an attraction pheromone.
This tick requires moisture and warmth, brush and bush and does not survive in open grassland. In South Africa it is found along the coastal belt from Port Elizabeth in the Eastern Cape Province, through KwaZulu-Natal and thence across Mpumalanga, Gauteng, Limpopo and North West Provinces, north of a line running approximately through Pretoria to the Botswana border. It is also present in eastern Swaziland, southern Mozambique, eastern Botswana and in southern and eastern Zimbabwe.
Laboratory studies have demonstrated that A. hebraeum can carry higher infections and also a greater number of strains of E. ruminantium than A. variegatum, the other major vector of this disease. In addition field observations seem to indicate that outbreaks of the disease are more commonly associated with the former than the latter tick and that these outbreaks are also more severe. Besides cattle a number of wild ruminants, as well as other animals such as scrub hares, helmeted guineafowls and leopard tortoises can act as asymptomatic carriers of E. ruminantium. Other organisms transmitted by A. hebraeum are Theileria mutans and Theileria velifera the cause of benign bovine theilerioses. The occurrence of foot abscesses in goats in the Eastern Cape Province, South Africa is significantly related to the seasonal abundance of adult A. hebraeum and Rhipicephalus glabroscutatum. The attachment sites of these ticks around and between the hooves afford entrance to secondary bacterial infections leading to abscessation. These abscesses cause severe lameness in the goats, particularly if more than one foot is affected.
Amblyomma variegatum – the tropical bont tick
Its common name is derived from its bright, contrasting colouration and the fact that it is common in the tropical regions of Africa. It transmits the agents of heartwater (E. ruminantium), benign bovine theilerioses (Theileria mutans, Theileria velifera), bovine ehrlichiosis (Ehrlichia bovis), the virus of Nairobi sheep disease and is associated with acute bovine dermatophilosis (Dermatophilus congolensis).

Amblyomma variegatum male |
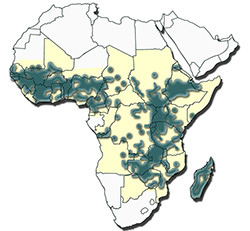
Distributiuon of Amblyomma variegatum |
It is widely distributed through West, Central, North-East and East Africa and in southern Africa extends into Zambia, north-eastern Botswana, the Caprivi Strip of Namibia, north-western Zimbabwe and central and northern Mozambique. Its spread southwards appears to be limited by a decrease in annual rainfall and interspecific competition with A. hebraeum with which it shares similar habitats, hosts and sites of attachment.
Vector-based epidemiology of heartwater (Norval et al. 1992)
Reservoirs of infection
African buffaloes and possibly other wild ruminants can serve as asymptomatic carriers of E. ruminantium, so can domestic ruminants. Repeated infection of hosts can reinforce the carrier state as the infectivity of immune hosts increases following re-exposure to the organism.
Non-ruminant animals such as scrub hares, guineafowls and tortoises are frequently infested by the immature stages of A. hebraeum, and can also be infected with E. ruminantium and can serve as reservoirs of infection for as yet undetermined lengths of time. Unfed, infected A. hebraeum nymphs and adults can probably retain infection for at least a year. Attached A. hebraeum and A. variegatum males can remain on the same host for 5 months or can transfer to other hosts. Nymphs do not lose the infection if they feed on non-susceptible hosts.
Acquisition of infection
All developmental stages of the tick can become infected while feeding on an infected host. Larvae only ingest small volumes of blood and are hence exposed to small numbers of organisms and do not seem to multiply the organisms effectively, hence larval to nymph transmission is not optimal. Nymphs can pick up low levels of infection and the organisms multiply effectively in their gut cells, and nymph to adult transmission is excellent. Adults can also become infected, but this is a dead-end infection in the females, unless they transfer to another host because transovarial transmission is very rare. Males that become infected can, however, remain attached for long periods of time and can transmit the infection repeatedly to the same host, thus boosting its carrier state, or they can transmit infection intrastadially should they transfer to another host.
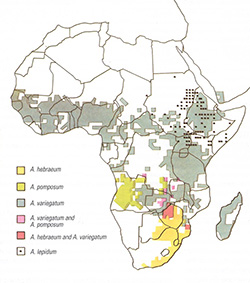
Distribution of the major Amblyomma spp. |
Vector status
Amblyomma hebraeum and A. variegatum are the most successful vectors as they feed on domestic ruminants in all their stages of development. It would, however, appear that the susceptibility to infection with E. ruminantium can vary considerably within populations of A. hebraeum and of A. variegatum. Field observations seem to indicate that outbreaks of the disease are more commonly associated with A. hebraeum than with A. variegatum, and that these outbreaks are also more severe. The parasite is passed to the host within the first 24 hours of tick attachment, and because the ticks are slow feeders they may still be attached when the animal shows the first signs of illness. The other Amblyomma spp. that can serve as vectors either have limited distribution ranges, or they do not feed on ruminants in all their stages of development, or they have very specific host preferences.
Factors leading to infection
Larvae ascend the surrounding vegetation and infest passing hosts. Nymphs and adults remain on the soil surface and are activated by specific stimuli, namely CO2 exhaled by host animals and by an attraction, aggregation, attachment pheromone produced by attached engorging male ticks. Thus herds of cattle are more likely to trigger tick activity than single animals, large animals more so than small animals, and already infested animals more so than uninfested animals. Most engorged larvae or nymphs detach from their hosts around sunset, and where animals are kraaled at night large accumulations of unfed nymphs or adults that have recently moulted from the detached engorged larvae and nymphs, will be present and these will have a readily available source of hosts in the kraaled cattle.
Control
The first effects of intensive tick control in endemic situations will be the reduction in size of the reservoirs of infection that reside in the ticks themselves. This will lead to a reduction in the numbers of animals that become carriers and the re-challenge of existing carriers will be lowered and this will lead to lower infection rates in ticks. The overall effect will be to reduce the numbers of animals that acquire immunity to heartwater through natural infection.
